|
Windows product keys are almost (but not quite) a thing of the past. With Windows 10, Microsoft has rewritten the rules for how it performs product activation on retail upgrades of Windows, including the free upgrades available for a year beginning on July 29, 2015. The net result is that clean installs will be much easier--but only after you get past the first one. OEM activation hasn't changed, nor have the procedures for activating volume license copies. 
But the massive means that for the near future at least those retail upgrade scenarios are very important. More Windows 10 • • • • • • • • The biggest change of all is that the Windows 10 activation status for a device is stored online. After you successfully activate Windows 10 for the first time, that device will activate automatically in the future, with no product key required. That's a huge change from previous versions of Windows, which required a product key for every installation. And it's potentially an unwelcome surprise for anyone who tries to do a clean install of Windows 10 without understanding the new activation landscape. I've had some way-off-the-record discussions with people who know a few things about the subject, and I've also done my own testing for the two weeks since Windows 10 was released to the public. Here's what I've learned. Your Windows 10 license is stored online and linked to your device. For more than a decade, one of the keys that Microsoft's activation servers have relied on is a unique ID, which is based on a hash of your hardware. That hash is reportedly not reversible and not tied to any other Microsoft services. So although it defines your device, it doesn't identify you. Here's how that ID works with Windows 7 or Windows 8: When you activate for the first time, that hashed value (let's call it your installation ID) is recorded in the activation database alongside the product key you entered with the installation. Later, when you reinstall the same edition of Windows on the same hardware, with the same product key, it's activated automatically. (Conversely, if you try to use that product key on a different machine with a different hardware ID, you'll probably be denied activation.) **Several people have asked about hardware upgrades. Windows 10 Product Key for Pro and Home Edition Free,Windows 10 activator, Windows 10 full download, Windows 10 iso file, Windows 10 license key. Feb 14, 2007 I lost my original dell computer after a power surge so I reformatted the hard drive and changed the motherboard. I reinstalled windows xp. Nov 20, 2012 Volume activation overview for Office 2010. A unique MAK key is given to an organization for each Volume License edition of Office 2010. See update at the end of this post. Windows 10 goes one very large step further. When you upgrade from Windows 7 or Windows 8.1, the Windows 10 setup program checks your current activation status and reports the result to the activation servers. If you're 'genuine' (that is, properly activated), the Windows activation server generates a Windows 10 license certificate (Microsoft calls it a 'digital entitlement') and stores it in conjunction with your installation ID and the version you just activated (Home or Pro). It didn't need a product key to do that activation. All it needed was the proof from the Software Licensing Manager utility that your underlying activation was legit. You can now wipe that hard disk completely, boot from Windows 10 installation media, and install a squeaky clean copy. The Setup program asks you to enter a product key, but in a major change from Windows 8 and 8.1, it allows you to skip entering that key. You'll be asked to enter that key a second time, later in setup, but you can skip past that box as well. When you finish the reinstall, assuming you used the same Windows 10 version on that hardware, you'll find it's automatically activated. I've tested this scenario on multiple machines, and the result has been consistent: Step 1: I booted from Windows 10 installation media, a USB flash drive prepared by the Windows 10 Media Creation tool, and tried a clean install on a system that had never been activated for Windows 10. I skipped both prompts to enter a product key. My system failed activation. Step 2: I reset the machine with its original, activated copy of Windows 7 or Windows 8.1 and then ran the Windows 10 online upgrade. At the end of the process, I confirmed that Windows 10 was properly activated. Step 3: I then wiped the hard drive clean and used the exact same media as in Step 1 to do a clean install of Windows 10. As before, I skipped the product key entry. I used a Microsoft account in one test and used a local account in another. After the installation was complete, the system showed that it had a properly activated copy of Windows 10. You can, of course, buy a full or OEM copy of Windows 10 on a flash drive, and you can also buy product keys online. You can use that product key to do a clean install on a system that has never run Windows 10 and it will get a license certificate from the activation servers. And just like those upgraded PC, it should then allow you to do a clean install of the same Windows 10 edition without having to re-enter the product key. For those who are anxious to do a clean install, here's the trick: Don't do the online upgrade. Instead, from your current, activated copy of Windows 7 or Windows 8.1, download the Windows 10 ISO file for the corresponding edition (Home or Pro), or create a bootable USB flash drive. Without exiting your current Windows version, double-click the ISO to mount it as a virtual DVD (or open the USB flash drive with installation media) and then double-click Setup. Your goal is to get to the screen with this full range of options. Windows 10 is a key part of Microsoft's plan to be more of an Internet of things player. The catch is that few people see Microsoft putting the pieces together. Choose the option I've highlighted at the bottom: the one that says you want to keep nothing. The Windows 10 Setup program installs a clean copy of the edition that corresponds to the one you have installed. As part of the process, it verifies the activation status of your old Windows, creates the new license certificate, and blows away your previous install. And you never had to enter a product key. After you restart, your clean copy of Windows 10 is activated, and you can reinstall it any time without having to worry about activation. And you'll never need a product key again. That's all well and good for those who are currently running Windows 7 or Windows 8.1. But what about those who did a clean install of a preview edition, never upgrading over a licensed copy? You can skip the product key during installation, but when you're done with Setup your system will be marked as not activated. You won't be able to use any personalization options, and you'll have a persistent watermark on the desktop warning you that you need to activate. To 'get genuine,' you're going to need to do one of two things: get a Windows 10 product key for the edition you have installed (you can use a key from MSDN or a retail source) or restore your old operating system, activate it, and then do the upgrade to register a license certificate. I honestly have no idea how the telephone activation hotlines will respond to calls from Insiders who want to activate a copy for the first time. This is new territory for Microsoft and for its customers. Update: In the comments, several people have asked what happens if you make changes to hardware. As I noted earlier, Microsoft doesn't provide details of how it calculates that hardware hash, but upgrades of system components such as a video card or a hard drive won't normally trigger a reactivation. If that happens, a quick call to the activation line will resolve the issue, often without any human contact required, in minutes. The one exception is a motherboard replacement, which will inevitably cause the Software Licensing Management utility to recognize the device as a new PC and require reactivation, typically over the phone. A motherboard upgrade, even if you reuse storage, video, memory, and a case, is considered a new PC. In that case, if the underlying Windows license is from a retail copy, that license can be transferred. If you are upgrading (and not replacing) a motherboard on an OEM PC that was sold with Windows preinstalled, the license agreement prevents the license from being transferred. By registering you become a member of the CBS Interactive family of sites and you have read and agree to the, and. You agree to receive updates, alerts and promotions from CBS and that CBS may share information about you with our marketing partners so that they may contact you by email or otherwise about their products or services. You will also receive a complimentary subscription to the ZDNet's Tech Update Today and ZDNet Announcement newsletters. You may unsubscribe from these newsletters at any time. ACCEPT & CLOSE.
0 Comments
Welcome to the unofficial community for discussing all-things. This subreddit is not affiliated with or monitored by Verizon Wireless or Verizon Communications. Oct 20, 2008 if you have the phone.keep it. Just activate it. Replacement phones return as often as verizon's. Many asurion replacement phones with. Many verified employees of Verizon Wireless and Verizon Wireless Premium and Authorized Retailers are active here. Verified employees have a flair next to their username. Rules • Please be respectful & don't be a dick. Being intentionally rude, condescending, or insulting will not be tolerated. Failure to comply with this rule will result in a ban without warning! • Please follow the. • Posts should be primarily about Verizon and their subsidiaries (Yahoo! Posts relating to a field of Verizon are also welcomed, but should be kept to a minimum. 
• Don't post any personal information, including your own. Similarly, do not send personal information by private message to anyone, even verified Verizon employees. • Before you post, to see if your question has already been answered. Many common questions are also answered in the, or linked directly from the sidebar. • Don't downvote a post or comment because you dislike it. Downvotes are only for when a post or comment is off-topic or not contributing to a on topic discussion. 
• Discussion about or encouraging use of illegal services on the Verizon network is not allowed. Post will be deleted. • Violating the Rules for the first time will incur a warning from the moderators. After your second offense, you will be temporarily banned. After your third offense, you will be permanently banned. If you have any questions regarding the Rules, please. Helpful Links Questions about Equipment Installment Plan (formerly ). If your new to Verizon & want to know how much your bill will be? You can use the,. Previous grandfathered unlimited data users can now upgrade to Verizon New Unlimited Data plans & save money. To be verified as an employee of Verizon Wireless or of a Premium or Authorized Retailer, with proof of employment. Verification is voluntary.: Verizon has an established Executive Support team on DSLR, for Wireline support. See for assistance. Also, visit,, and, for general discussion. Verizon Wireless Contact • Phone: (800) 922-0204 or *611 from any Verizon Phone • • • Social Media: & Coverage Maps • • • • LTE Band Information • Device Subreddits • • • •. I left my G4 in a taxi last night. No big deal, that's what insurance is for, right? The following evening I file a claim with Asurion. After I finish filing it, I head to the Verizon store to activate my old Droid X2 for a few days until I get my replacement. Once I have a working phone, I find out that someone not only found my phone, but wants to get it back to me! So, I call up Asurion to try and get my claim canceled so I can save the $150 deductible. This is less than one hour after I finished filing the initial claim. I'm told that since the claim has already been filed, I can't cancel it, even though it hasn't shipped yet. They tell me that I have to mail the phone back to them and they'll start to process my refund. So, the question is, why shouldn't I just sell the refurbished phone that they'll send me? I'll probably make more than the deductible, and I don't have to worry about paying for shipping, or tracking the phone and keeping my fingers crossed that the refund actually comes through. This information pertains to customers that have replaced their lost or stolen phone with a new phone/device. To activate your phone/device, please follow the instructions provided in the box with your new device. If the instructions were misplaced, please use the following steps to activate your phone. Note: If you suspended your service due to a vacation, you should contact Customer Care to reactivate your line. For more information about suspending or re-instating your service, see the support page. 1) Please make sure you install the new SIM card that was provided in your box. 2) Verify that the new SIM card is properly installed in your device, and that your device has been fully charged. 3) After installing the new SIM card correctly and fully charging the device, turn the device on and place a test call. If you are able to make and receive calls on your phone, your device is activated. If you are unable to make calls and you have an acceptable signal to place and receive calls, please continue with the next steps. 4) Call 1-866-895-1099 or visit. 5) Please have the following information available: Your wireless phone number, billing ZIP code, and your social security number or tax identification number. 6) You will be prompted to agree to AT&T's Terms & Conditions by providing an electronic signature (over the phone or online). 7) Within 20 minutes, your handset should be working. If you continue to have issues activating your phone, please call 1-866-391-0749. 
CiteSeerX - Scientific documents that cite the following paper: A spreading activation theory of semantic processing. It is assumed that activation is very rapidly spread over a semantic network. The ACT* spreading-activation theory predicts an. The spread of activation. Learn about how knowledge is organized in the mind. Created by Carole Yue. Watch the next lesson: Missed the previous lesson? MCAT on Khan Academy: Go ahead and practice some passage-based questions! About Khan Academy: Khan Academy offers practice exercises, instructional videos, and a personalized learning dashboard that empower learners to study at their own pace in and outside of the classroom. We tackle math, science, computer programming, history, art history, economics, and more. Our math missions guide learners from kindergarten to calculus using state-of-the-art, adaptive technology that identifies strengths and learning gaps. We've also partnered with institutions like NASA, The Museum of Modern Art, The California Academy of Sciences, and MIT to offer specialized content. For everyone. Subscribe to Khan Academy’s MCAT channel: Subscribe to Khan Academy. Lexical Decision (Editor: ) Introduction How is our knowledge organized? Early theorists (e.g., Collins & Loftus, 1975) suggested that knowledge is organized within semantic networks, an array of nodes representing concepts and links that transmit activation between nodes. In such a conceptualization, if the node representing 'pencil' was activated, the activation would spread to nodes representing related items. As a result, after hearing or seeing the word 'pencil,' people would retrieve or recognize words such as 'paper' or 'pen' faster than they would retrieve or recognize words such as 'sleep' or 'queen,' because concepts related to pencil have already been activated. Many experiments have demonstrated that such facilitation, called priming, does indeed occur. Interest in network models was refined with the introduction of parallel distributed processing models, illustrative of a connectionist perspective, that McClelland and Rumelhart (1986) popularized. Connectionism applied physiological principles about neural circuits, excitatory and inhibitory connections, and distributed coding (the idea that a concept is represented by a pattern of activation, rather than by activation of a single node) to concept representation. Although some connectionist principles are controversial, distributed memory models inform explanations of semantic priming (Masson, 1995). Meyer and Schvaneveldt (1971) provided early evidence supporting network models by illuminating the effects of priming. In their classic experiment, these researchers measured response times as people made lexical decisions (determining whether or not two letter strings, presented simultaneously, were both words). In conditions in which both stimuli were words, some of the pairs were related (e.g., BREAD and BUTTER) and others were unrelated (e.g., CHAIR and FLOWER). The key finding from this investigation was that response time was faster for related words than for unrelated words, consistent with the concept of spreading activation. Beyond studies of accessing individual words from our mental lexicon (dictionary), investigators have applied the time course of spreading activation to explain individual differences in general comprehension skills. For example, to determine how people with differing comprehension skills processed meanings of ambiguous words (e.g., 'bugs' might mean either 'hidden microphones' or 'insects'), Gernsbacher and Faust (1991) presented a sentence, followed by either immediate or delayed presentation of an ambiguous word. Findings indicated that all participants experienced interference with no delay: however, after a brief delay, people with good comprehension skills more effectively suppressed activation of inappropriate meanings than did people with weaker comprehension skills. Researchers continue to study how a variety of variables influence performance on lexical decision tasks. For example, Grieco, Betella, Conti, Orioli, and Casco (2004) documented that, although lexical decision time is typically slower for longer words than for shorter words, even if a preview of the word is provided at the point of fixation, this word length effect is absent if a preview of the word appears in a peripheral location. Additionally, Schilling, Rayner, & Chumbley (1998) reported that word frequency effects were greater in lexical decision tasks than in naming tasks or measures of eye fixation times. The present experiment was designed both to replicate and extend the original Meyer and Schvaneveldt (1971) investigation. Although the present task is similar to that of Meyer and Schvaneveldt (indicating if two letter strings are both words or non-words), the present experiment introduces two other variables. First, the interstimulus interval between the two letter strings varies (permitting assessment of the time course of activation). Second, some of the non-words were intentionally designed to closely resemble words that relate to the actual words with which they are paired. By presenting word-like stimuli, we can examine the extent to which non-words might facilitate recognition of actual words that may be interpreted as related to the non-words. Design The experiment uses a 2 (Stimuli type: word or non-word) x 2 (Stimuli relationship: related or unrelated) x 3 (Interstimulus interval: 300, 600 or 900 msec.) repeated-measures design, with 12 conditions. Word stimuli were selected based on norms developed by Nelson, McEvoy, and Schreiber (1998), considering both associative strength and frequency. Paralleling the Meyer and Schvaneveldt procedure, your task is to look at two letter strings and decide, as quickly as possible, whether both letter strings are words or non-words. You may respond either by clicking 'word' or 'non-word,' or by using the up and down arrow keys. 
You will complete 72 trials, 6 in each condition. Data Analyses Data is downloadable in three formats (XML, Excel spreadsheet format, and comma delimited for statistical software packages). Figure 1 shows an excerpt from a sample Excel spreadsheet. The first five columns provide classification data (participant ID number, class id, gender, age, and completion date. The next 12 columns indicate the response times in seconds for each of the interstimulus intervals for the Related Words, Unrelated Words, Related Non-Words, and Unrelated Non-Words. For example, the first set of data columns provides average response times for Related Words cues at interstimulus intervals of 300, 600 and 900 milliseconds. 
Figure 1 A simplified analysis targeted at examining the basic effect of word association on lexical decision time would involve only the word conditions. Thus, you would conduct a 2 (Stimuli relationship: related or unrelated) x 3 (Interstimulus interval: 300, 600 or 900 msec.) repeated-measures factorial ANOVA, with lexical decision time as the dependent variable. You would expect to find a main effect of stimulus relationship, such that lexical decision time would be faster for related words than for non-related words. You might also examine whether this effect is more pronounced at different interstimulus intervals. If the analysis revealed a stimuli relationship x interstimulus interval interaction, you would then use posthoc tests to determine at which interstimulus interval the effect of stimuli relationship was strongest, providing insight into the time course of spreading activation. A more complete analysis would also include the non-word conditions, requiring a 2 x 3 x 2 (Stimulus type: word or non-word) repeated-measures factorial ANOVA. Such an analysis would reveal if the effect of stimulus relationship was similar for words and non-words. If that were to occur, this complete analysis would also reveal a main effect of stimulus relationship (with lexical decision time being faster for related words than for nonrelated words) and no stimuli type x stimuli relationship interaction. In contrast, if the relatedness effect occurred only for words but not for nonwords, the analysis would reveal a stimulus type x stimulus relationship interaction. This complete analysis would also permit you to explore whether or not there was an effect of interstimulus interval on lexical decision time and if the time course of activation differed for words and nonwords. You would use post hoc tests to more fully illuminate the nature of any significant interactions. Applications/Extensions More recent studies have explored the neural correlates of priming in lexical decision tasks. For example, using functional magnetic resonance imaging (fMRI), Rossell, Bullmore, Williams, and David (2001) identified brain areas that differently responded to lexical decisions involving automatic (no delay) and controlled (longer delay) processing and other brain areas that differently responded to primed and unprimed lexical decisions. In another fMRI study involving primes that had multiple meanings (e.g., 'bank'), Copland, de Zubicaray, McMahon, and Eastburn (2007) determined that different neural areas were involved in the processing of lexical decisions for target words associated with dominant (e.g., money) or subordinate (e.g., river) meanings. Lexical decision tasks are a common tool for identifying processing similarities and differences between special populations and control groups. For example, based on performance in a lexical decision task with pseudohomophones (non-words, e.g., snoe or werd, that are pronounced similarly to real words), Transfer and Reitsma (2005) inferred that deaf and hearing children both used phonological (acoustic) encoding when they were reading. Crosbie, Howard, and Dodd (2004) found that children with specific language impairment performed less accurately than children without impairment on an auditory lexical decision task, although the speed of responses was similar for both groups. Other research has indicated that speed of lexical access was slower for patients with Parkinson's Disease than for normal controls, but was affected by whether or not patients had taken dopamine as medication (Angwin, Chenery, Copland, Murdoch, & Silburn, 2007). However, Blum and Freides (1995) determined that thought-disordered schizophrenic patients performed similarly to a control group without schizophrenia when making lexical decisions. Overall, lexical decision research with special populations illuminates the extent to which semantic processing is impaired in the target groups. References Angwin, A. J., Chenery, H. J., Copland, D. A., Murdoch, B. E., & Silburn, P. The speed of lexical activation is altered in Parkinson's disease. Journal of Clinical and Experimental Neuropsychology, 29, 73-85. A., & Freides, D. Investigating thought disorder in schizophrenia with the lexical decision task. Schizophrenia Research, 16, 217-224. M., & Loftus, E. A spreading-activation theory of semantic processing. Psychological Review, 82, 407-426. A., de Zubicaray, G. I., McMahon, K., & Eastburn, M. Neural correlates of semantic priming for ambiguous words: An event-related fMRI study. Brain Research, 1131, 163-172. L., Howard, D., & Dodd, B. Auditory lexical decisions in children with specific language impairment. British Journal of Developmental Psychology, 22, 103-121. Gernsbacher, M. A., & Faust, M. The mechanism of suppression: A component of general comprehension skill. Journal of Experimental Psychology: Learning, Memory, and Cognition, 17, 245-262. Grieco, A., Bettella, S., Conti, M., Orioli, M., &Casco, C. The effect of preview eccentricity in a lexical decision task. Visual Cognition, 11, 781-796. A distributed memory model of semantic priming. Journal of Experimental Psychology: Learning, Memory, and Cognition, 21, 3-23. McClelland, J. L., Rumelhart, D. E., & Hinton, G. The appeal of parallel distributed processing. Rumelhart, J. McClelland, & the PDP research group (Vol. Eds.), Parallel distributed processing: Explorations in the microstructure of cognition: Vol. Cambridge, MA: MIT Press. E., & Schvaneveldt, R. Facilitation in recognizing pairs of words: Evidence of a dependence between retrieval operations. Journal of Experimental Psychology: General, 90, 227-234. L., McEvoy, C. L., & Schreiber, T. The University of South Florida word association, rhyme, and word fragment norms. Retrieved August 6, 2007 from University of South Florida Free Association Norms Web site: Rossell, S. L., Bullmore, E. T., Williams, S. C., & David, A. Brain activation during automatic and controlled processing of semantic relations: A priming experiment using lexical-decision. Neuropsychologia, 39, 1167-1176. Schilling, H. H., Rayner, K., & Chumbley, J. Comparing naming, lexical decision, and eye fixation times: Word frequency effects and individual differences. Memory & Cognition, 26, 1270-1281. Transler, C., & Reitsma, P. Phonological coding in reading of deaf children: Pseudohomophone effects in lexical decision. British Journal of Developmental Psychology, 23, 525-542. • • • In association with:,,. All materials on this site are protected by United States Copyright Law and may not be reproduced, distributed, transmitted, published or broadcast without prior written permission. You may not alter or remove any trademark, copyright, or other notice from copies of the content. The OPL web site managed by the OPL Advisory Board and materials remain the property of the authors. All Rights Reserved. Funding provided by the National Science Foundation [DUE - 0435058]. Adobe CS3 Master Collection ISO Serial Key Free Download Adobe CS3 Master Collection ISO Full Version: Adobe CS3 Master Collection ISO Serial Key Free Download Latest Version for Windows. It is full offline installer standalone setup of Adobe CS3 Master Collection ISO. Adobe CS3 Master Collection ISO Overview: Adobe CS3 Master Collection is really a handy suite that has got just about all Adobe software programs like Adobe InDesign CS3, Adobe Photoshop CS3, Adobe Expensive, Adobe Dreamweaver, Adobe Fireworks, Adobe Acrobat, Adobe Consequences etc. You may also download Adobe InDesign CS6. Adobe InDesign CS3 is desktop posting software that you can use for creating posters, flyers magazines and newspapers etc. It’s the first version which assists Apple based Apple computers and table styles. Adobe Illustrator most likely the favourite application currently that you can use for editing your photos and you may turn your regular photo right into a masterpiece. Adobe Illustrator CS3 has faster startup occasions but got helpful Quick Selection tool. 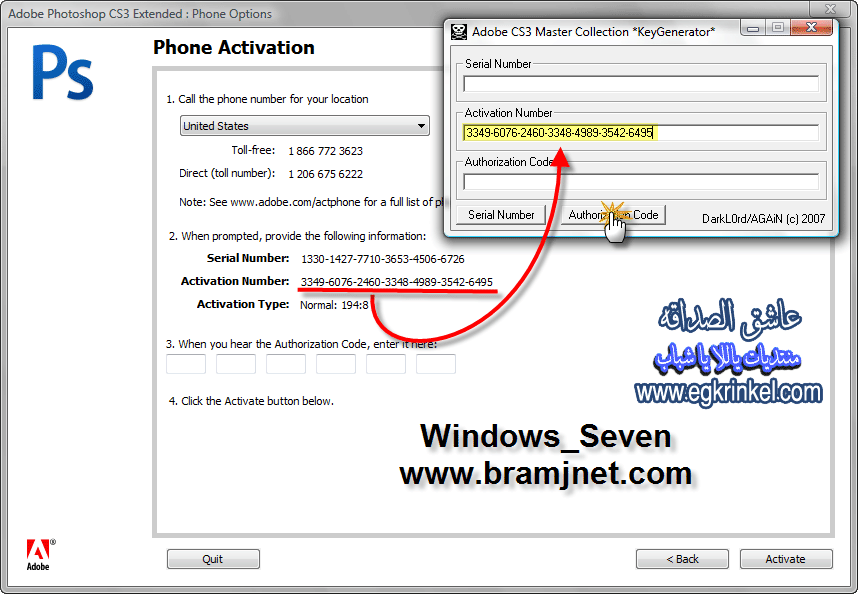
Adobe Dreamweaver is yet another application within this master collection pack that you can use for developing websites easily and in an exceedingly a shorter period. Should you not have enough coding understanding than Dreamweaver may be the application for you personally. Adobe Fireworks CS3 is really a graphic editor that you can use for editing graphics inside a website and Adobe Consequences is really a video editor which is going to do miracles for you personally. Aug 30, 2017 Properly Re-Activate Adobe CS3, CS4, CS5 Now re-launch your Adobe app, like Photoshop, and you should should be greeted with a proper activation. You may also download Adobe Dreamweaver CS3. Adobe CS3 Master Collection Serial Key Free Download Languages Mac OS X Windows English—International (4.4 GB) (323 MB) (1.0 MB) (3.0 GB) (259 MB) Japanese (4.4 GB) (461 MB) (1.0 MB) (2.9 GB) (365 MB) Dutch French French—Canada German Italian Portuguese—Brazil Spanish Spanish—Latin America Spanish—North America (4.3 GB) (342 MB) (1.0 MB) (2.8 GB) (269 MB) The software applications included in this collection are • Adobe InDesign® CS3. • Adobe Photoshop® CS3 Extended. • Adobe Illustrator® CS3. • Adobe Flash® CS3 Professional. • Adobe Dreamweaver® CS3. • Adobe Contribute® CS3. • Adobe Fireworks® CS3. • Adobe After Effects® CS3 Professional. • Adobe Premiere® Pro CS3. • Adobe Soundbooth® CS3. • Adobe Encore CS3. • Adobe Acrobat® 8 Professional. • Adobe Bridge CS3. • Adobe Stock Photos. • Adobe Device Central CS3. • Adobe Acrobat Connect™. • Adobe Dynamic Link. • Capture In After Effects. • Adobe OnLocation™ CS3. • Adobe Ultra® CS3. Features of Adobe CS3 Master Collection ISO Serial Key Free: Below are some noticeable features which you’ll experience after Adobe CS3 Master Collection ISO free download. • Handy suite which has got all Adobe software application. • Got Adobe InDesign CS3 by which you can create posters and magazines. • Can edit your photos with Adobe Photoshop CS3. • Can develop websites with Adobe Dreamweaver CS3. • Can edit graphics in a website with Adobe Fireworks CS3.  
• Can edit videos with Adobe After Effects CS3. Adobe CS3 Master Collection ISO Technical Setup Details: • Software Full Name: Adobe CS3 Master Collection ISO Serial Key Free • Setup File Name: Adobe_CS3_Master.iso • Full Setup Size: 3.3 GB • Setup Type: Offline Installer / Full Standalone Setup • Compatibility Architecture: 32 Bit (x86) / 64 Bit (x64) • Latest Version Release Added On: 25th Jan 2016 • License: Free Trial • Developers: System Requirements For Adobe CS3 Master Collection ISO Before you start Adobe CS3 Master Collection ISO free download, make sure your PC meets minimum system requirements. • Operating System: Windows XP/Vista/7/8/8.1/10 • Memory (RAM): 1GB of RAM required. • Hard Disk Space: 3.31GB of free space required. • Processor: Intel Pentium 4 or later. Adobe CS3 Master Collection ISO Free Download Click on below button to start Adobe CS3 Master Collection ISO Free Download. This is complete offline installer and standalone setup for Adobe CS3 Master Collection ISO. This would be compatible with both 32 bit and 64 bit windows.  If you're unable to download the latest version of Internet Explorer, please ensure you have compatibility view turned off. How to disable compatibility view • Open Internet Explorer. • Click on the Tools menu (you may have to press Alt to bring up the menu). • If the Compatibility View box is checked, click to remove checkmark.  
Start using your smartphone with TELUS. It's easy to activate online and choose the plan that's right for you. Activate your phone. Get your phone going. It's easy to activate your phone online. Plus you can activate a TELUS phone, or one that's unlocked and compatible. New customers. Dial 1-888-552-3333. • Click Tools again. • Select Compatibility View Settings. • Make sure 'rogers.com' is not listed under 'Websites you've added to Compatibility View.' • Close and reopen Internet Explorer. • Go to rogers.com. The original owner will know if it's locked or not. Have them call Telus and get the phone unlocked. Chances are that Telus will do this for free (as the CRTC has ruled that no carrier locks are allowed as of Dec 1 of this year). Test it with your Rogers SIM before you give them the cash. If it says 'Invalid SIM' then it's still locked on Telus and you should hold off on paying them until it's unlocked. (if they can't get it unlocked by Telus, it could even be a stolen device - and blacklisted - so it may not work on any network). Yep, the Apple Store is a great suggestion. This isn't entirely accurate however: ' If the phone is locked to Telus, you would need to have the original owner unlock it by Telus and then give it to you.' Any Telus subscriber with a device locked to Telus could get the device unlocked (provided there isn't still subsidy associated with that phone) - if so the subsidy would need to be paid off before they would agree to unlock it. They need to have their active Telus SIM in the device when they call to request the removal of the SIM lock. Hello This is the accurate information from their website. The owner much either have it registered the device for 90 days on the account if it was done via upgrade or if the device balances is not owing anymore or they bought it outright it can happen immediately. That's all I am saying. So no one can just go to Telus, get a sim card and ask then to unlock it other then the original owner. ' To be eligible to have your subsidized device unlocked, your current device must have been active on the TELUS network for a minimum of 90 days. There is a one-time fee for unlocking. If you have purchased your phone outright or do not have an outstanding device balance, you can pay a one-time fee to immediately unlock your device. Eligibility Both your device and your account must meet all eligibility conditions below. TELUS may refuse any unlock request that appears intended to defraud TELUS or its customers. And, how does that differ from what I said? ' Your account is associated with the device to be unlocked' - means that the device has been used by the current owner.not necessarily that they are the original owner. If there's no subsidy on the device and it's not stolen, any Telus subscriber with an active subscriber SIM in the device could have it unlocked. (the call centre rep will look to see which device is connected by the subscriber at the time of the call). You just like being argumentative or quoting regulations? Abstract Posttranslational modifications of histones, alterations in the recruitment and functions of non-histone proteins, DNA methylation, and changes in expression of noncoding RNAs contribute to current models of epigenetic regulation. Nuclear receptors (NRs) are a group of transcription factors that, through ligand-binding, act as sensors to changes in nutritional, environmental, developmental, pathophysiologic, and endocrine conditions and drive adaptive responses via gene regulation. One mechanism through which NRs direct gene expression is the assembly of transcription complexes with cofactors and coregulators that possess chromatin-modifying properties. Chromatin modifications can be transient or become part of the cellular “memory” and contribute to genomic imprinting. Because many food components bind to NRs, they can ultimately influence transcription of genes associated with biologic processes, such as inflammation, proliferation, apoptosis, and hormonal response, and alter the susceptibility to chronic diseases (e.g., cancer, diabetes, obesity). The objective of this review is to highlight how NRs influence epigenetic regulation and the relevance of dietary compound–NR interactions in human nutrition and for disease prevention and treatment. Identifying gene targets of unliganded and bound NRs may assist in the development of epigenetic maps for food components and dietary patterns. Progress in these areas may lead to the formulation of disease-prevention models based on epigenetic control by individual or associations of food ligands of NRs. Introduction Nuclear receptors (NRs) are a group of transcription factors that, through ligand binding, drive adaptive gene responses to changes in nutritional, environmental, developmental, pathophysiologic, and endocrine conditions (). The NR superfamily includes endocrine, adopted orphan, orphan with evidence of natural and/or synthetic ligand, and orphan with no known ligand NR subclasses. Endocrine NRs bind the ovarian hormone receptors estradiol (ER) and progesterone, androgen receptor (AR), vitamin D receptor (VDR), retinoic acid receptor (RARα, RARβ, RARγ), retinoid acid receptor [retinoid X (RXRα, RXRβ, RXRγ)], glucocorticoid receptor (GR), mineralcorticoid receptor, and thyroid hormone receptor. The adopted orphan NRs are mediators of metabolic pathways and bind lipids [PPARα, PPARβ/δ, PPARγ liver X receptor (LXRα, LXRβ)] and bile acids [BAs; farnesoid X receptor (FXR)]. The orphan NR subclass, for which there is evidence of natural and/or synthetic ligands, includes the small heterodimer partner (SHP) and aromatic hydrocarbon receptor (AhR). The SHP binds retinoid-related molecules (, ) and regulates BAs and lipid homeostasis (). The AhR binds dietary (e.g., resveratrol, indol-3-carbinol), endogenous (e.g., PGs), and synthetic polycyclic aromatic hydrocarbon (PAH), dioxin (e.g., 2,3,7,8-tetrachlorodibenzo- p-dioxin), and polychlorinated biphenyl compounds (). Orphan NRs with no known ligands include the dosage-sensitive sex reversal-adrenal hypoplasia critical region on chromosome X, gene 1 ( DAX1), whose mutation predisposes to X-linked adrenal hypoplasia congenital and hypogonadotropic hypogonadism (). The DAX1 protein is a corepressor of FXR. Overexpression of DAX1 downregulates the expression of FXR target genes and interferes with BAs, TGs, and glucose metabolism (). Epigenetic regulation by nuclear receptors. Nuclear receptor activity DNA methylation. Polycomb complex and gene activation through. It is well accepted that epigenetic mechanisms, i.e., changes in gene expression without modification in nt sequence, such as DNA methylation, histone modifications, and noncoding mRNAs, establish areas of active or repressed chromatin, which mediate adaptive responses to dietary, environmental, and developmental signals (). Research evidence suggests that dietary compounds contribute to epigenetic regulation and phenotypic plasticity through both NRs and nongenomic pathways. For example, the soy compound genistein stimulates gene expression through induction of phosphoinositide 3-kinase/AKT, which in turn phosphorylates and represses the histone methyltransferase (HMT) enhancer of zeste homolog 2 (EZH2), thus reducing the amounts of the histone 3 trimethylated at lysine 27 (H3K27me3) repressive mark (). In parallel, genistein induces transcription of estrogen-responsive genes by inducing the recruitment of ERα and cofactors with histone-modifying properties (e.g., p300) (). Similarly, by engaging both cytosolic (i.e., extracellular signal-regulated kinase 2) and nuclear (i.e., RXR) mediators, retinoic acid modifies the epigenetic landscape and transcriptional regulation of genes involved in differentiation and embryonic development (, ). Moreover, a large number of dietary ligands influence the epigenetic machinery by acting as methyl donors, cofactors, coenzymes, and regulators of chromatin-modifying proteins (). 
Therefore, the full impact of diet–NR interactions on health and disease can only be appreciated by developing models that integrate all the pathways that impinge on epigenetic regulation (). This review will focus on how the interplay between food compounds and NRs influences epigenetic regulation and the relevance of these interactions for disease prevention and treatment. NR Structure and Regulation The general structure of NRs comprises an amino-terminal activation function domain-1 (AF-1), a DNA-binding domain (DBD), a hinge region, a ligand-binding domain, and a C-terminal activation function domain-2 (AF-2) (). The ligand-binding domain comprises coregulator interaction domains. Through the DBD, NRs can bind directly to DNA as monomers, homodimers, or heterodimers (reviewed in references and ). For example, the ERα forms homodimers at estrogen response elements, whereas the FXR can form heterodimers with the RXR at FXR response elements (FXRE). 
Certain NRs function as promiscuous partners for different receptors. This is the case for the RXR, which forms complexes at target promoters with the PPAR, VDR, RAR, and FXR. In addition, NRs can modulate gene expression through physical interactions with DNA-bound transcription factors. For example, the ERα and ERβ can enhance or repress, respectively, transcription of genes through interactions with DNA-bound activator protein-1 (AP-1) complexes () (). FIGURE 1 Nuclear receptor structure and coregulation by food ligands and nutritionally related xenobiotics and drug compounds. NRs share a general DNA structure comprising an amino-terminal AF-1, an H, an LBD, and a C-terminal AF-2 domain ( A). Examples of NR interactions within the coregulator interaction domains: the ERα forms homodimers at EREs; the ERα and ERβ can enhance or repress, respectively, transcription of genes containing AP-1 sites through interactions with DNA-bound Jun/Fos complexes; the AhR/ARNT heterocomplex binds DNA at XREs; the FXR forms heterodimers with the RXR at FXRE ( B). Certain NRs function as promiscuous partners for various receptors. This is the case for the RXR that forms complexes at target promoters with FXR and PPAR, VDR, and retinoic acid receptor. Examples of food ligands are listed below each NR. The diagram depicts general models of repression and activation via CpG methylation and demethylation mediated by NRs ( C). 
In the absence of ligands or when NRs are bound to antagonists, NRs are found in NCoR complexes with factors that possess DNMT and HDAC, HMT, and histone demethylase (e.g., LSD-1) properties. Certain covalent modifications placed by NR corepressors on histones are markers of silenced heterochromatin and include H3K9me3, histone 3 trimethylated at lysine 27, and H4K20me3. Chromatin is occupied by the repressive HP-1 factor. Binding of food ligands to NRs triggers the dismissal of NCoR complexes and the recruitment at the target promoter of NCoA complexes that comprise HMT (e.g., MLL-3) and HAT (e.g., cAMP-response element-binding protein/p300) factors. The latter possess enzymatic activities that place “active” histone acetylation marks on histones 3 and 4 (H3K9Ac, H4K20Ac). The physical interaction between NRs and cofactors influences the magnitude and direction of the transcriptional response (, ). For instance, transcription by the ER, RAR (), and PPARγ () is potentiated by interactions with the cofactors p160/steroid receptor coactivator-1 (SRC-1) and cAMP-response element-binding protein (CBP)/p300 through LXXLL-binding motifs (in which L indicates leucine and X indicates any amino acid) (). Moreover, coregulator exchange contributes to gene-, cell-, and tissue-specific transcriptional regulation by NRs. For example, LXRα-regulated lipogenesis and cholesterol/BA homeostasis in the liver requires binding of LXRα through LXXLL motifs with the NR coactivator 6. However, NR coactivator 6 is not required for regulation of transcription by the ERα in the mammary gland (). Conversely, repression of transcription by unliganded NRs is achieved through interactions with LXX I/H I XXX I/L motifs on nuclear corepressor (NCoR) and silencing-mediator for retinoic and thyroid (SMRT) factors (). NRs can also modify transcriptional regulation through interactions with cis-acting transcription factors. For example, the looping of the cytochrome P450, family 1, subfamily A, polypeptide 1 ( CYP1A1) promoter region around histones facilitates interactions of DNA-bound AhR/AhR nuclear translocator (ARNT) complexes with the specificity protein 1 transcription factor recruited at adjacent sites and enhances transcription of the CYP1A1 gene in response to AhR ligands (, ). Examples of cooperativity between NRs and cis-acting transcription factors include those between the DNA-bound GR and the accessory nuclear factor-1 on the 11β-hydroxysteroid dehydrogenase () and phosphoenol-pyruvate-carboxykinase ( PEPCK) genes (). An example of negative cis-interaction is the recruitment of the activated AhR to a xenobiotic response element [XRE (5′-GCGTG-3′)] harbored in the breast cancer-1 ( BRCA-1) gene, which displaces an ERα/p300 complex from an adjacent AP-1 site and represses estrogen-dependent activation of BRCA-1 expression (). NRs may also repress transcription without direct binding to DNA through competition for ubiquitous cofactors as in the case of the trans-RA/RAR/RXR heterocomplex, which interferes with AP-1-dependent trans-activation by excluding the cofactor CBP (). Similarly, the SHP orphan NR, which lacks a DBD, represses transcription through direct interaction with AF-2 coactivator domains on NRs or by stimulating the recruitment of NCoRs (). Moreover, signaling pathways impart on NRs and cofactors posttranslational modifications (e.g., phosphorylation, methylation, and acetylation) that influence transcriptional activity. For instance, phosphorylation of p160 potentiates its coactivator functions on the AR and ER (). Similarly, methylation and p300-dependent acetylation of the ERα protein potentiate its transcriptional activation of estrogen-responsive genes (). The cyclical recruitment of NRs and cofactors at target promoters contributes to transcriptional control (). This is the case for the VDR on the 25-hydroxy vitamin D3 1α-hydroxylase ( 25OH-D3-1α) (), the AR on the prostate specific antigen (), the PPARδ on the pyruvate dehydrogenase kinase 4 ( PDK4) (), and the AhR on the CYP1A1 () and cyclooxygenase-2 ( COX-2) () genes. Similarly, ligands of the AhR direct the cyclical recruitment of p300 to the COX-2 gene and its association with acetylated (Ac) histone 4 (H4) (). Fluctuations in NR and cofactor recruitment fine-tune the duration of activation (or repression) and transcriptional rates of target genes. Thus, modeling the impact of food ligands for NRs on epigenetic regulation of target genes would require knowledge of the following: 1) the presence and spatial arrangement of core binding motifs for NRs and cofactors; 2) kinetics of recruitment of NR complexes; and 3) the effects on expression and posttranslational modification of NRs and related cofactors. Epigenetic repression In the absence of ligands or when NRs are bound to antagonists, NRs are found in corepressor complexes with factors that possess histone deacetylase (HDAC), HMT, histone demethylase (HDM), and phosphatase enzymatic activities () (). The NR cofactors Sin3 transcription regulator family member A (Sin3A), NCoR, and SMRT lock target genes in a repressed state by forming bridges with HDACs. NR corepressor complexes include the ATP-dependent switch/sucrose non-fermentable (SWI/SNF) nucleosome remodeling factors. Certain covalent modifications placed by NR corepressors on histones are markers of silenced heterochromatin and include histone 3 trimethylated at lysine 9 (H3K9me3), H3K27me3, and H4K20me3. The binding of heterochromatin protein-1 to H3K9me3 locks heterochromatin in a transcriptionally silent state (). The transrepressive functions of certain NCoR complexes (e.g., tailless-like X receptor) require the presence of the HDM lysine-specific demethylase-1, which demethylates H3K4me2 and H3K4me (). Competition among NRs for coregulators with histone-modifying properties (e.g., p300, SRC-1) is a mechanism that contributes to epigenetic repression. For example, by “squelching” p300 from ERα complexes, the agonist-bound AhR represses transcription of DNA repair (e.g., BRCA-1) () and other estrogen-inducible genes (). Similarly, the binding of vitamin D to VDR causes the dissociation of p300 and the recruitment of HDAC corepressor complexes on the 25OH-D3-1α gene, hampering expression of 25OH-D3-1α and its participation in the de novo synthesis of vitamin D (). Ubiquitin-mediated degradation of NCoRs is another mechanism that contributes to epigenetic repression by NRs (). For example, agonist binding increases the affinity of PPARγ for NCoR/HDAC-3 repressor complexes and prevents ubiquitin-dependent removal of NCoR and transcriptional activation of the NF-κB-inducible proinflammatory COX-2 gene (). Epigenetic activation For many genes, binding of agonists to NRs triggers the dismissal of NCoRs and the recruitment to the target promoter of coactivator complexes containing CBP and p300. The latter possess histone acetyltransferase (HAT) activities that place acetylation marks on H3 and H4 (H3K9Ac, H4K20Ac) (). For example, transcriptional activation of the type 1 keratin KA11 ( KA11) gene by IL-1β is accompanied by dismissal of NCoR from the AR bound to the KA11 promoter () Similarly, the estradiol-bound ERα induces transcription of the human pS2 ( pS2) gene via dismissal of NCoRs and the ordered recruitment of activator complexes () comprising SRC-1 and HATs. These factors facilitate the acetylation and dimethylation of H3K14 and histone-4 at arginine-3 (H4R3) and the recruitment of transcription binding proteins and polymerase II, leading to activation of the pS2 gene. Histone modifications associated with transcriptional activation by NR include methylation of H3K4 (H3K4me) by the H3K4-methyltransferase mixed lineage leukemia-3 (, ) and phosphorylation of H3 at serine 10. The later interferes with the binding of heterochromatin protein-1 to H3K27me3 and promotes nucleosomal opening and transcriptional activation (). For example, elevation of phosphorylation of H3 at serine 10 and H3K9Ac are observed during early phases of ERα-induced transcription initiation (). The binding affinity of ligands toward NRs influences the dynamics of chromatin modifications at target genes. A study that examined the modulation of PPARγ transcriptional activity by insulin-sensitizing thiazolidinedione (TZD) compounds reported that pioglitazone and rosiglitazone were more effective than troglitazone in inducing the recruitment of PPARγ complexes to the PEPCK and PDK4 promoters and acetylation of PEPCK- and PDK4-associated histones (e.g., AcH4) (). The stronger transcriptional effects of pioglitazone and rosiglitazone were mainly attributed to their higher binding affinity for PPARγ. Conversely, the preventive effects of resveratrol against transcriptional repression by the bound AhR of estrogen-inducible genes were related to formation of resveratrol/AhR heterocomplexes with lower affinity for XRE () and the agonist actions of resveratrol on the ERα () and related cofactors (e.g., p300) (). Although the promiscuity of NRs for ligands with diverse binding affinity offers great variability and adaptability to environmental challenges, it also greatly expands the opportunities for the development of NR-based therapeutic strategies with food ligands (). Cytosine-phosphate-guanine (CpG) methylation and demethylation at target promoters Hypermethylation of cytosines comprised in CpG islands and its propagation through DNA replication have been related to gene silencing, whereas demethylation of CpGs has been linked to gene activation (). Enzymes implicated in maintenance and de novo DNA hypermethylation are, respectively, DNA methyltransferase (DNMT)-1, and DNMT-3a and DNMT-3b, respectively (). DNMT-1, but not DNMT-3a and DNMT-3b, and methylated cytosine binding protein-2 (MeCP2) recruit the biotin protein ligase holocarboxylase synthetase (HLCS) (), leading to the assembly of a multiprotein gene repression complex comprising HDAC-1, NCoR, and the eukaryotic histone methyl transferase-1 (EHMT-1). The binding between HLCS and EHMT-1 is facilitated by the HLCS-dependent biotinylation of K161 in EHMT-1 (). The close proximity between HLCS and histones facilitates the HLCS-dependent biotinylation of histone H3 and H4 (). Nevertheless, biotinylation of histones is a rare event (≤0.001% of histone H3 and H4) and does not account for the repressive role of biotin on gene expression (–), in particular of repeats, which appears to be caused primarily by the HLCS-containing multiprotein complex (). Repression of repeats is necessary for maintenance of genome stability, whereas chromosomal abnormalities have been reported in biotin-depleted human cell cultures (). DNMTs physically bind to HMTs and HDACs, providing a mechanistic link between transcriptional repression via DNA hypermethylation and histone methylation and deacetylation. In the case of the ERα-inducible pS2 gene, cycles of CpG methylation alternate with cycles of CpG demethylation at the pS2 promoter. The hypermethylation of CpGs occurs at the end of each productive transcription cycle and corresponds to the corecruitment of MeCP2, SWI/SNF, DNMT-1, and DNMT-3a/3b. Conversely, transcriptional activation by the ERα, GR, and VDR has been linked to stimulation of CpG demethylation at the pS2 (, ), tyrosine aminotransferase (), and cytochrome P450, family 7, subfamily B, polypeptide 1 () promoter, respectively. Whereas the mechanisms of DNA methylation have been established clearly, the processes that lead to active DNA demethylation remain an area of active investigation (). Previous models of demethylation included the following: 1) the enzymatic removal of the methyl group from 5-methylcytosine; 2) nt excision repair involving the growth arrest and DNA damage 45 (GADD45) protein and XPG, a 3′ endonuclease; 3) direct base excision repair (BER) of 5-methylcytosine by DNA glycosylases [i.e., thymine DNA glycosylase (TDG)] and methyl-CpG binding domain-containing protein 4 (MBD4); 4) and deamination and repair of 5-methylcytosine via TDG and BER to replace the mismatched T with a C (, –). Evidence that GADD45 family members interact with many food-related NRs, such as RXRα, RARα, ERα, PPARα, PPARβ, and PPARγ, suggest that nt excision repair-based CpG demethylation plays an important role in epigenetic regulation by common dietary compounds. However, the role of GADD45 in demethylation is still debated, and mounting evidence now offers a model for a complete cycle of methylation and demethylation that involves 4 steps: 1) CpG methylation by DNMTs; 2) iterative oxidation of methylated cytosine by 10-11 translocation enzymes; 3) excision of methylated cytosine by TDG to generate an abasic site; and 4) placement of unmodified cytosine by BER (–). Evidence that TDG interacts with RAR, RXR, and ERα (–) further highlights the role of food-related NRs in epigenetic control via CpG demethylation. PPAR PPARα, PPARβ, and PPARγ play a key role in FA and glucose metabolism. PPARα is a binding target for several FAs, including palmitic acid, oleic acid, linoleic acid, CLA, arachidonic acid, EPA, DHA, and the PG metabolite 15D-J2 () (). PPARα is expressed primarily in liver, heart, and skeletal muscle. Gene targets for PPARα encode enzymes that contribute to FA oxidation: 1) acyl-CoA synthetase; 2) carnitine palmitoyl-transferase-1; 3) acyl-CoA oxidase; 4) very-long-chain and medium-chain acyl-CoA dehydrogenase; and 5) FA transport protein. Also, PPARα activates its own expression through binding to a hepatocyte nuclear factor-4α-binding element harbored in the PPARα promoter (). Gestational exposure to a protein-restricted diet elevates PPARα expression by lowering PPARα promoter CpG methylation in hepatic () and heart () tissue of rat offspring. Conversely, maternal supplementation with folic acid induces hypermethylation of the PPARα gene in liver tissue of offspring (). These observations suggest that maternal nutrition influences epigenetic regulation of PPAR-regulated pathways in offspring. Nonalcoholic fatty liver disease is a condition characterized by increased lipid influx into the liver and de novo hepatic lipogenesis. The hepatic accumulation of TGs has been linked to increased association of the repressive histone mark H3K9me3 on the PPARα gene and lowering of PPARα expression (). Methyl-donor deficiency is a condition that has been related to impaired FA oxidation and decreased expression of PPARα. For example, administration during pregnancy and lactation of a methionine- and choline-deficient diet led to hypomethylation of the PPAR coactivator 1α (PGC-1α) protein and its reduced stimulation of PPARα expression in liver tissue of rat offspring (). Thus, dietary disturbances (e.g., methyl-donor deficiency) may contribute to the pathogenesis of fatty liver by reducing expression of PPARα through epigenetic mechanisms. Conversely, targeting of PPARα with the synthetic agonist 2-[[4-chloro-6-[(2,3-dimethylphenyl)amino]-2-pyrimidinyl]thio]acetic acid (WY14,643) has been proposed as an option for the management of hypertriglyceridemia. The treatment with WY14,643 ameliorated liver insulin resistance, exerted anti-inflammatory effects, and activated β-oxidation enzymes in a rodent model of hepatic and muscle steatosis (). Unfortunately, the benefits of lipid-lowering interventions based on WY14,643 and other agonists of PPARα could be offset by increased cancer risk. For example, increased PPARα expression was correlated with tumor development in humans, raising some concerns that constitutive hyperactivation of PPARα may exert tumor-promoting effects (). In a rodent model (SV129 mice), the long-term exposure (5 mo) to WY14,463 increased liver cell proliferation and global hypomethylation but decreased the amounts of the repressive H4K20me3 mark. The latter changes were not observed in SV129/ PPARα −/− mice. These data suggested a positive role for PPARα-mediated epigenetic regulation in tumorigenesis (). As a result, more experimental evidence is needed to establish whether or not therapies based on dietary or synthetic agonists of PPARα pose increased cancer risks to humans through the induction of undesirable epigenetic changes (). In mammals, white adipose tissue (WAT) plays a prominent role in storing excess energy in the form of TGs but also plays essential roles as an endocrine organ through secretion of leptin and adiponectin and metabolism of sex steroids and glucocorticoids (). Resident macrophages in WAT are a meaningful source of inflammatory cytokines, such as TNF-α and IL-6. An increase in circulating amounts of these macrophage-derived factors in obesity leads to a chronic low-grade inflammatory state that has been linked to the development of insulin resistance and diabetes (). There are 2 known PPARγ splice variants, PPARγ1 and PPARγ2. Expression of the PPARγ1 isoform is higher in WAT, intestine and spleen. PPARγ2 is expressed preferentially in WAT and brown adipose tissue (BAT). The activation of PPARγ with the synthetic agonist rosiglitazone shifts the fate of pluripotent adipose-derived stem cells toward BAT (). In addition, manipulation of PPARγ signaling in WAT produces gene expression representative of BAT and improves glucose tolerance in murine cell cultures and in mice, respectively (). Ligands of PPARγ comprise common dietary FAs (linoleic acid, arachidonic acid, EPA, DHA), metabolites (15d-PGJ2), synthetic TZDs, and nonsteroidal anti-inflammatory drugs (, ). Genes induced by PPARγ include those that regulate FA (e.g., FA-binding protein and lipoprotein lipase) and carbohydrate ( PEPCK) metabolism (). Chromatin-modifying cofactors that colocalize with PPARγ comprise p300/CBP, SRC-1, and jumonji histone demethylase 2A (JHDM2A), an HDM. Histone marks associated with activation of transcription by PPARγ are H3K4me2, H3K4me3, H4K20me1, acH3K9ac, and acH3K27. Conversely, the association of PPARγ with HDACs (HDAC-1 and HDAC-3), nuclear corepressors (NCoR-1, NCoR-2, and SMRT), and HMTs [SET domain, bifurcated 1 (SETDB1) and histone-lysine-N-methyltransferase (SUV39H1)] has been linked to transcriptional repression. Repressive histone marks associated with promoter recruitment of PPARγ include H3K9me2, H3K9me3, H3K27me2, and H3K27me3 (). The epigenetic silencing of the PPARγ gene by MeCP2- and EZH2-containing repressive complexes is a biomarker of colorectal cancer progression and adverse patient’s outcome (). Hepatic reactivation of PPARγ expression via repression of EZH2 and MeCP2 functions has been documented for the dietary compounds rosmarinic acid and baicalin (). Intrauterine growth restriction is a condition that, in neonatal rat lung, reduces expression of PPARγ by lowering the amounts of the HMT su(var)3-9, enhancer of zeste, trithorax, domain 8 (setd8) and H4K20me on the PPARγ gene. The latter effects are ameliorated in offspring by maternal supplementation with DHA (). A factor related to PPARγ that controls lipid metabolism is the PPARγ coactivator PGC-1α. In skeletal muscle, the acute exposure to a heavy lipid load (e.g., palmitate) reduces PGC-1α expression through DNMT-3b-dependent non-CpG methylation of the PGC-1α promoter and impairs FA oxidation (). Conversely, exercise induces acute expression of PGC-1α, PDK4, and PPARδ in skeletal muscle via reduced CpG methylation of the respective promoters (). This cumulative evidence illustrates the contribution of epigenetic modification through DNA methylation at the PGC-1α gene in the regulation of lipid metabolism by FAs and exercise in skeletal muscle. TZDs are a group of insulin sensitizers, of which, troglitazone induces transcription of the PPARγ2 gene through placement of the active H4K20me1 mark on the PPARγ2 promoter (). The induction of expression of PPARγ2 is linked to CpG demethylation of the PPARγ2 promoter in differentiated 3T3-Ll adipocytes (). Similarly, the transcriptional activation of the glucose-dependent insulin-tropic polypeptide receptor and PEPCK genes with synthetic ligands of PPARγ is related to increased association of AcH3 and AcH4, respectively, with the glucose-dependent insulin-tropic polypeptide receptor and PEPCK promoters (, ). Because certain TZDs, such as rosiglitazone, induce severe health side effects, such as myocardial infarction (, ), future studies should examine whether or not feeding of certain food compounds alone or in combination with antidiabetic drugs offer safer alternatives for epigenetic activation of PPARγ networks and lowering of circulating glucose in patients with diabetes. ER The ER is expressed in 2 isoforms, α and β. The ERα stimulates proliferation in reproductive organs (). The estradiol-bound ERα recruits cofactors that possess HAT (p300, p160s), HMT (e.g., protein arginine methyltransferase 1), and HDM (e.g., lysine-specific demethylase-1) enzymatic activities. Conversely, ERα bound to antagonists (e.g., tamoxifen) associates with NCoR-1 and SMRT. The latter recruit nucleosome remodeling and deacetylase complexes that repress transcription. Amounts of NCoR-1 are usually lower in invasive breast cancers, implying that changes in epigenetic control linked to NCoR-1/HDACs may contribute to transition to more invasive phenotypes (). The activation of ERβ leads to transcriptional repression of ERα target genes through either the sequestration of cofactors for the ERα or formation at target genes of complexes that repress ERα-dependent transcription. For example, the genistein-bound ERβ triggers the recruitment of the cofactor GR interacting protein-1, which antagonizes the activation of ERα responsive promoters (). The contribution of ERβ to epigenetic regulation appears to be gene and tissue specific. In differentiated mouse embryonic fibroblasts, ERβ induces hypomethylation of the glucose transporter 4 (GLUT4) promoter and restores GLUT4 gene transcription (). Conversely, in prostate cancer cells, ERβ stimulates the recruitment of corepressor complexes containing NCoR, mSin3A, and HDAC-1 and the CpG methylation of the glutathione S-transferase P1-1 promoter (). Isoflavones are a class of phytoestrogens with higher (∼10-fold to 30-fold) binding affinity for ERβ compared with ERα (e.g., genistein > biochanin A > daidzein). The phytoalexin resveratrol also binds to the ERα and ERβ but with ∼7000-fold lower affinities compared with estradiol (). Through epigenetic mechanisms, isoflavones exert tumor-protective or -promoting effects in ER-positive mammary tissue depending on the timing of exposure. In rodent models, the prenatal and neonatal exposure to genistein increased the incidence of carcinogen-induced mammary tumors () and uterine adenocarcinoma (), respectively. The tumor-promoting effects of neonatal exposure to genistein in uterine tissue have been related to constitutive stimulation of hypomethylation and expression of the nucleosomal binding protein-1 ( NSBP-1) gene (). Notably, the NSBP-1 gene encodes for a member of the high-mobility group nucleosome-binding proteins that reduce compaction of chromatin and enhance transcription. The NSBP-1 protein is expressed at high amounts in cancer cells, whereas the knockdown of the NSBP1 gene inhibits tumor growth in nude mice but induces G 2/M cell cycle arrest and apoptosis (). In adult life, genistein may increase breast cancer risk through epigenetic silencing of RARβ2. The latter mediates the anticancer effects of retinoic acid. DNA hypermethylation of the RARβ2 gene was observed in nipple aspirates obtained from premenopausal women supplemented for 1 menstrual cycle with phytoestrogens (90.6 mg/d genistein, 36.4 mg/d daidzein, and 1.8 mg/d glycitein) and plasma amounts of genistein >600 μg/L (). The DNA methylation rate of RARβ2 is generally higher in breast tumors and precancerous breast lesions compared with normal breast tissue (). Some of the preventive effects of genistein and daidzein against ERα-positive breast cancer have been related to activation of BRCA-1 expression via reversal of CpG methylation at the BRCA-1 gene (). Genistein mimics the stimulatory effects of estradiol on BRCA-1 (, ) and triggers the association of p300, SRC-1, and AcH4 with the BRCA-1 promoter (, ). Moreover, genistein may exert anticancer effects in endocrine tissue through epigenetic reactivation of ERα () and repression of DNMT-3b expression (). The antidiabetic effects of daidzein and its metabolite equol have been attributed to the induction of GLUT4 expression by ERβ through reduced CpG methylation at the GLUT4 promoter (). Similarly, studies that focused on the anticancer effects of the phytoalexin resveratrol reported that it reduced promoter methylation of the phosphatase and tensin homolog gene in ERα-positive MCF-7 breast cancer cells (, ). Overall, these examples suggest a role for polyphenols, such as genistein, resveratrol, and daidzein, as modifiers of cancer and diabetes risk through epigenetic regulation of genes targeted by the ER. AhR Ligands of the AhR include environmental dioxins and PAHs, a multitude of dietary compounds (e.g., resveratrol, kaempferol, and indol-3-carbinol), and metabolites (i.e., PGs) (reviewed in references, ). In the absence of ligands, the AhR is found in the cytoplasm in association with the chaperone proteins heat shock protein 90, HBV X-associated protein 2 (XAP2), and p23. The binding of ligands to the AhR displaces XAP2 and favors migration of the bound AhR to the nucleus in which it interacts with ARNT, of which 3 different isoforms (ARNT-1, ARNT-2, and ARNT-3) have been identified. After nuclear release of heat shock protein 90, the AhR/ARNT heterocomplex binds to core XREs and activates the expression of phase I and phase II enzymes. Phase I enzymes comprise CYP1A1, cytochrome P450, family 1, subfamily B, polypeptide 1, and cytochrome P450, family 1, subfamily A, polypeptide 2, which produce chemically reactive species. The latter are substrates for detoxification by phase II enzymes, such as NAD(P)H:quinine oxidoreductase and UDP-glucuronosyltransferase-1A6 (). The role of CpG methylation in differential regulation of CYP1A1 and cytochrome P450, family 1, subfamily B, polypeptide 1 expression by the AhR has been documented clearly in breast and hepatic cells (). In addition to its putative role in detoxification, the AhR regulates transcription of estrogen-responsive genes (). For example, in the absence of agonists, the AhR coactivates the ERα and is necessary for estrogen-dependent activation of BRCA-1 transcription (). Conversely, in the presence of agonists (i.e., PAHs, dioxins), the bound AhR is recruited to XREs harbored in the BRCA-1 promoter and antagonizes ERα-dependent stimulation of BRCA-1 expression. The latter effect is paralleled by increased CpG methylation of the BRCA-1 promoter and association of HDAC-1, DNMTs (DNMT-1, DNMT-3a, and DNMT-3b), and H2K9me3 with the BRCA-1 gene (). Among many dietary ligands of the AhR, resveratrol is a prototype food compound that overrides the placement of repressive marks by AhR agonists on the BRCA-1 gene (). VDR Through epigenetic mechanisms, the VDR directs the expression of genes involved in vitamin D and bone tissue remodeling. The cholecalciferol, 1,25-dihydroxycholecalciferol, [1,25(OH)2-D3]-bound VDR heterodimerizes with RXR at VDR response elements [(A/G)G(G/T)TCA] and recruits SWI/SNF chromatin remodeling (e.g., BRG-1) and HAT factors (e.g., CBP/p300, SRC-1) that activate transcription of the 25-hydroxy vitamin D3-24-hydroxylase (cytochrome P450, family 24, subfamily A, polypeptide 1) and osteopontin genes (). The cytochrome P450, family 24, subfamily A, polypeptide 1 enzyme and osteopontin maintain vitamin D and bone tissue homeostasis, respectively, through the conversion of 25OH-D3 and 1,25(OH) 2-D3 into hydroxylated degradation products and stimulation of bone cell survival. Conversely, the recruitment of the RXR/VDR–1,25(OH) 2-D3 heterocomplex to a VDR-interacting repressor bound to an E-box (CANNTG) harbored in the cytochrome P450, family 27, subfamily B, polypeptide 1 (CYP27B1) promoter represses CYP27B1 transcription (). The CYP27B1 enzyme catalyzes the 1α-hydroxylation of vitamin D3. The transrepression of CYP27B1 transcription by the VDR/VDR-interacting repressor heterocomplex involves the recruitment of HDACs, Sin3A, NCoR, DNMTs, and MBDs on the CYP27B1 gene. The CpG demethylation and derepression of the CYP27B1 promoter is induced by the parathyroid hormone through PKC-dependent phosphorylation of the DNA glycosylase MBD4 and BER mechanisms (, ). Therefore, dietary ligands of VDR coordinate calcium and bone tissue metabolism by altering the epigenetic code of genes involved in vitamin D metabolism. FXR Endogenous ligands of FXR are the primary BAs chenodeoxycholic and cholic acid and the secondary metabolites deoxycholic acid and lithocholic acid. The chenodeoxycholic acid is the BA with the highest binding affinity for FXR (EC 50 of 10–50 μmol/L). Conversely, the absence of a 7α-hydroxy group in deoxycholic acid and lithocholic acid or placement of a 12α-hydroxy group in CA reduces the binding affinity of these compounds for FXR, which directs transcription as a monomer, homodimer, or through heterodimerization with RXR. The activated FXR represses transcription of genes involved in BA synthesis (cytochrome P450, family 7, subfamily A, polypeptide 1) and gluconeogenesis (glucose-6-phosphatase). Conversely, it enhances the transcription of genes involved in BA shuttling [ileal bile acid-binding protein and metabolism ( SHP)] (reviewed in reference ). Reduced FXR expression is found in intestinal epithelial cells with activated Wnt (wingless-type MMTV integration site family) signaling (), human colon carcinoma (), rodent colorectal cancers carrying a mutated adenomatous polyposis coli gene, and humans with familial adenomatous polyposis. FXR induces the expression of tumor suppressor ( p21) and proapoptotic (FA synthase) but represses expression of antiapoptotic (B-cell CLL/lymphoma 2) and TNFα, genes. Also, FXR tethers the transcription factor p65, thus hampering the ability of NF-κB to activate proinflammatory (e.g., COX-2 and inducible nitric oxide synthase) genes (). The SHP and the bile salt export pump genes are targets for epigenetic regulation by the FXR. Under conditions of low exposure to BAs, the FXR is deacetylated by sirtuin 1, and transcription of SHP is repressed. Conversely, the activation of FXR by BAs induces the coordinate recruitment of p300 to the FXR/RXR complex, release of sirtuin 1, and association of AcH3K9 and AcH3K14 with the SHP promoter (, ). Similarly, FXR activation with the synthetic compound 3-[2-[2-chloro-4-[[3-(2,6-dichlorophenyl)-5-(1-methylethyl)-4-isoxazolyl]methoxy]phenyl]ethenyl]benzoic acid (GW4064) increases the association of AcH3K9 and AcH3K14 with the SHP promoter (). FXR-dependent transactivation of the bile salt export pump gene requires histone methylation by the H3K mixed lineage leukemia-3 () and H4R3 protein arginine methyltransferase 1 () methylases. A posttranslational modification of FXR that increases binding of the FXR/RXR complex to FXRE and expression of FXR target genes is methylation of lysine 26 by the methylase histone-lysine N-methyltransferase SET domain 7/9 (SET7/9) (). The latter enzyme also methylates the ERα at lysine 302 (). These data clearly corroborate the role of epigenetic mechanisms in FRX-dependent transcriptional regulation of genes involved in maintenance of BA homeostasis. Significance and Future Perspectives The field of epigenetics finds its roots in the pioneering theories of Conrad Hal Waddington () and others (), who developed the concept that the epigenotype is the sum of the mechanisms that bridges the gap between genotype and phenotype (). Research findings discussed in this review highlighted the notion that NRs influence the epigenotype, and thus the phenotypic response, by directing gene expression through the recruitment to target promoters of factors that possess chromatin, histone, and DNA-modifying properties. The ability of NRs to affect gene expression via epigenetic mechanisms would be sequence specific, i.e., via direct binding to core elements (i.e., XRE, VDR response element, FXRE, etc,) or to factors (i.e., AP-1, specificity protein 1) bound to DNA motifs. The precise sequence of the core motifs and their spatial arrangement would determine the specificity, strength, and impact of NRs–response element interactions on gene expression (). Therefore, the identification and cataloging of binding sequences would greatly improve our understanding of how unliganded and food-bound NRs contribute to the epigenotype associated with chronic diseases. Notably, NRs interact promiscuously with many food ligands (). Therefore, future research should examine how synergies and antagonisms between food components and drugs for NRs influence the epigenotype and response to disease therapy. For example, studies reported that the efficacy of breast cancer therapies based on the ERα antagonist tamoxifen was augmented by the food isoflavone and ERα ligand genistein (). Conversely, the treatment with the PPARα agonist fenofibrate prevented glucocorticoid-induced hyperinsulinemia of mice fed a high-fat diet but potentiated the anti-inflammatory effects of the GR on NF-κB. These examples provide the rationale for the development of combination therapies for the management of chronic disorders, such as breast cancer and chronic inflammation (). Hence, future research should help clarify why diseased cells/tissues that share the same genotypes with healthy ones have different epigenotypes and predict their response to therapies based on food ligands of NRs. Finally, it is relevant to highlight that epigenetic events, such as DNA methylation, may be imprinted through somatic transmission or even exert transgenerational effects if they affect the germ line (). Nevertheless, unlike genetic changes (i.e., mutations), DNA methylation is reversible. For example, reversal of DNA hypermethylation has been documented for genistein on the RARβ () and ERα () and for epigallocatechin 3-O-gallate on the RARβ () genes. Whereas the anticancer effects of genistein and epigallocatechin 3-O-gallate may be due to combinatorial actions on NRs and other factors that influence epigenetic control, we suggest that vast opportunities exist for nutritional therapy of disease-related epigenotypes using food ligands of NRs. Footnotes • 1 Supported by U.S. Army Medical Research and Materiel Command Department of the Army Medical Division grant 10-1-0215 (to D.F.R.), American Institute for Cancer Research grant 10A058 (to D.F.R.), Arizona Cancer Center Support grant P30CA23074, and NIH grant R01 DK063945 (to J.Z.). • 2 Author disclosures: D. Romagnolo, J. Zempleni, and O. Selmin, no conflicts of interest. No More Mixing Bleach! Automatically Mixes a 10% Solution Every Time! • Convenient Activate Bleach Sprayer automatically mixes and dispenses 5.25% sodium hydrochlorite (bleach) with tap water at a 9:1 ratio of water to bleach, to produce a 5000 ppm bleach disinfectant and sterilant for institutional use on hard, non-porous surfaces • EPA registered broad-spectrum disinfectant bleach is approved to kill C. Activate™ Bleach Dilution System, Deardorff Fitzsimmons Supplier: DEARDORFF FITZSIMMONS CORP. The sprayer keeps the bleach separate from. Features of the Activate Bleach Dilution System: # Sprayer System automatically mixes and dilutes 5.25% sodium hypochlorite with tap water to make a 5000 ppm bleach. Activate™ Bleach Dilution System Sprayer & Water Bottle| Convenient sprayer system automatically mixes and dispenses the CDC-recommended 10% sodium hypochlorite disinfectant solution (5000 ppm available chlorine) for hospital and laboratory use on hard, non-porous surfaces. Fast and effective against C.difficile. Disinfection/Sterilization > Surface Disinfectants > Activate Bleach Sprayer with spray head, reusable water cartridge (left) and disposable bleach cartridge (right) Activate Bleach Sprayer Starter Kit includes 2 spray heads, 2 reusable water cartridges and 6 disposable bleach cartrdige refills Activate™ Bleach Sprayer No More Mixing Bleach! Automatically Mixes a 10% Solution Every Time! • Convenient Activate Bleach Sprayer automatically mixes and dispenses 5.25% sodium hydrochlorite (bleach) with tap water at a 9:1 ratio of water to bleach, to produce a 5000 ppm bleach disinfectant and sterilant for institutional use on hard, non-porous surfaces • EPA registered broad-spectrum disinfectant bleach is approved to kill C.   
Fay has two documents linked to each other—a form linked to an information document. On her work PC when she hovers the mouse pointer over the link it shows as a 'pointer hand.' On her PC at home she doesn't get the hand, but can right-click and select to open the link. Fay is wondering why the behavior is different on the two systems and what she can do to fix her home PC. 
Once you have installed Microsoft Word 2007 on your computer, you can run the program 25 times without activating it. Word then switches to Reduced Functionality mode. Adobe Acrobat X Accessibility Accessible PDF from Microsoft Word 3 Word 2007 Headings • Select the Home Ribbon in Word 2007 and select the proper heading. 
The behavior difference that Fay notes is actually controlled by a configuration setting within Word. Click the Office button and then choose Word Options. Click Advanced and display the Editing Options portion of the options. (This is the options at the very top of the advanced options.) (See Figure 1.) Figure 1. Advanced options in Word. Pay attention to the setting named Use CTRL+Click to Follow Hyperlink. If this check box is selected then Word requires the use of the Ctrl key to activate the link. (Or, as Fay did, right-clicking the link and selecting to open it.) If the check box is unselected then you'll get the 'pointer hand' and be able to simply click on a link to follow it.  
Product keys overview Visual Studio subscriptions provide access to a wide range of Microsoft products for designing, developing, testing, and demonstrating applications. Some of these products require product keys during installation, and some of those require activation. To create more accurate search results for Visual Studio 2003 try to exclude using commonly used keywords such as: crack, download, serial, keygen, torrent, warez. Jan 19, 2016. Total downloads: 144,013 Rank: 5 in C/C Last Updated: May 13, 2013 License: Freeware Free OS: Windows Vista/2003/XP Requirements: No special. Please be aware that Brothersoft do not supply any crack, patches, serial numbers or keygen for Visual C 2008 Express Edition,and please consult.   
Visual Studio subscriptions typically include five product keys for current versions of Windows and Office products, and three keys for older versions. Most product keys also allow multiple activations of the product for each key. Many products also allow you to install and use the product for 30 days or more without activation. For most subscribers, this provides more than enough activations to meet their needs. However, if you have exhausted the available activations for a product, you can submit a request for additional keys through Visual Studio. Activating a product Activation is required for certain products. Generally, in Visual Studio subscriptions this is limited to Windows, Windows Server, Microsoft Office client applications, and Expression Studio. When activation is required, many products — including Windows and Office — allow you to install and use the product without activation for 30 days or more. If you re-image your computers frequently (at least once every 30 days), consider NOT activating the product. Product keys supplied as part of your Visual Studio subscription do not allow unlimited activations of a product. If you’ve claimed all the available product keys under your subscription and have used up all the available activations, you can request additional keys by contacting your local Visual Studio Subscription. Requests will be considered on a case-by-case basis. If approved, product keys will be accessible in. Retail and Volume Licensing versions of products Visual Studio subscriptions offer a selection of retail and Volume Licensing versions of Microsoft products. Certain products are available from Downloads only in their retail version or only in their Volume Licensing version; some are available in both versions. Generally, products requiring activation, such as Windows, Windows Server, and Office client products, are only offered in their retail version through Downloads. Volume Licensing customers can also download software from the Volume Licensing Service Center (VSLC) to get Volume Licensing versions of products (and the corresponding product keys). Access to the VLSC is managed by designated administrators within the customer’s organization. Daily product key claim limits Microsoft is committed to protecting the integrity of the Visual Studio subscriptions program and the value of the software and benefits that the program’s legitimate subscribers receive for their investment. In support of this commitment, Microsoft has implemented daily key claim limits for Visual Studio subscriptions. Daily key claim limits restrict the number of keys that can be claimed per day per subscription. The daily key claim limit is 10 keys for all Visual Studio subscriptions, including those offered through MPN, BizSpark, MCT Software & Services, and MCT, with the exception of Visual Studio Professional (MPN), which has a daily key claim limit of 7 keys. Note: Key claim limits are subject to change. Please check here for the most up-to-date limits. If you have claimed the maximum number of keys available for your subscription within a 24-hour period, you will see an error telling you that your limit has been reached. The limit resets every 24 hours, starting at 12:01 AM Pacific Time. Our research shows that most subscribers are covered by the number of product keys included in their subscription. If you need additional keys, you can submit a request through Visual Studio Subscription and it will be considered for approval on a case-by-case basis. Locating and claiming product keys You must be signed in to your Visual Studio subscription to view your product keys. Individual product keys are found by selecting the blue Get Key link for a particular product on the page as shown below. All keys are also available in aggregate on the page. When multiple keys exist for a single product, notes will be displayed on the Notes column for the download to assist you in identifying which key should be used. Some products bundle multiple editions of the product into a single download. In these cases, the product key entered determines which edition of the product is installed. Some keys are provided automatically, such as “static” keys, which you can use as many times as needed because activation is not required. Other keys must be claimed by selecting the Get Key link for the product. A variety of key types are available, depending on the product. Product key types Key Type Description Not Applicable No key is needed to install this product. Retail Retail keys allow multiple activations and are used for retail builds of the product. In many cases, 10 activations are allowed per key, though often more are allowed on the same machine. Multiple Activation A Multiple Activation Key (MAK) enables you to activate multiple installations of a product with the same key. MAKs are generally used with Volume Licensing versions of products. Typically, only one MAK key is provided per subscription. Static Activation Key Static activation keys are provided for products that do not require activation. They can be used for any number of installations. Custom Key Custom keys provide special actions or information to activate or install the product. VA 1.0 These are multiple activation keys, similar to a MAK. OEM Key These are Original Equipment Manufacturer keys that allow multiple activations. DreamSpark Retail Key These retail keys are for DreamSpark and allow one activation. DreamSpark Retail keys are issued in batches and are primarily intended for student consumption. DreamSpark Lab Key These lab use keys are for DreamSpark programs and allow multiple activations. DreamSpark Lab Keys are intended for use in university computer lab scenarios. DreamSpark MAK Key These are MAK keys for DreamSpark program customers. You can claim a key from the download page for the product, or you can search for the key you need on the page. Claiming product keys Only subscribers with active subscriptions can download products and claim product keys. You can export your claimed keys from the page while your subscription is active. To claim a product key: • Sign in to your Visual Studio subscription. You must be signed in to download products or claim product keys. • Click on the tab. • Product keys are listed alphabetically by the name of the product. You can either scroll down to the name of the desired product, or search for it using the search bar at the top of the page. In this example, we’ve used the search bar to locate a product key for Visio 2010. As you can see, there are several versions of Visio 2010 listed. One key each has already been claimed for Visio Standard 2010 and Visio Premium 2010, and both have four keys remaining. The keys that were claimed are both retail keys, and are displayed on the page. Notice that you can record a brief note about claimed keys in the Notes column. You can use this in conjunction with the date in the Claimed column to keep track of the keys you’ve claimed. You might, for example, make notes when you activate an installation of the product using the key. Exporting your claimed keys You can export a list of all the keys you have claimed, along with a large selection of static and other keys that are automatically marked as “claimed” for you. IMPORTANT: If your subscription expires, you will no longer be able to claim new keys or export your claimed keys. To export your keys, simply click on the Export all keys button at the far right of the Product Keys page. An.xml file entitled KeysExport.xml will be created, and you’ll have the option to open or save the file. You will need to open the file with an application capable of handling.xml files. For example, you can open the file as a read-only workbook in Excel. Internet Demonstrations via Terminal Services With a Visual Studio subscription, you are allowed to provide end users access to Internet demonstrations of your programs via Terminal Services (Windows Server 2003 or Windows Server 2008) or Remote Desktop Services (Windows Server 2008 R2 and later). Up to 200 anonymous users can simultaneously access your demonstration this way. Your demonstration must not use production data. Visual Studio subscribers are licensed to demonstrate their applications to end users. This Internet demonstration using Terminal Services (TS) or Remote Desktop Services (RDS) is the only scenario where end users without a Visual Studio subscription can interact with the demonstration application when the software is licensed through Visual Studio subscriptions. This is in addition to dev/test rights, where Visual Studio subscribers can use as many RDS or TS connections as needed. Enabling RDS Access Visual Studio subscribers can increase the number of users who can access a Windows Server via RDS by entering a product key supplied in the tab on the. To obtain a product key, connect to the Product Keys page and scroll down to the version of Windows Server you are running. Locate “Windows Server R2 Remote Desktop Services connections” and click the Claim Key link. For example, if you’re using RDS on Windows Server 2012 R2 and your deployment uses user CALs, choose “Windows Server 2012 Remote Desktop Services user connections (50)”. Five keys of each type are available for Windows Server 2008 R2, and each key will support 20 connections. For Windows Server 2012 R2, four keys for each type are provide and will support 50 connections each. To enable additional connections in Windows Server: • Open Server Manager. • Open the Servers list in the left nav pane. • Right click on your license server and choose “Install Licenses”. • Follow the steps in the wizard. When you’re selecting the agreement type, choose “License Pack (retail)” and enter the product key you obtained from the MY portal. End users can connect to access applications via RDS if the following conditions are met: • Users must be anonymous (in a non-authenticated state). • Connections must be over the Internet. • Up to 200 concurrent user connections may be used for demonstrations of the application. • The product keys to enable user connections must be obtained by a Visual Studio subscriber. If you need instructions for setting up RD Licensing on your server, please see. If you have any questions, please visit the. MY Main PC crashed hard (motherboard) so I reloaded an Older testbed PC, until I get a new PC up and running. I reloaded up XP and added my Office 2003 Pro, when I tried to Activate the product I needed to use the telephone support. The message for CANADA said that telephone support has ended for this product. Is this the future of Microsoft, Deactivating the ability to reload older products!!!!! And forcing you to buy new. (I hate Win 8, reminds me of Vista and Millennium) The only reason I bought the office pack was for Outlook and Power Point, I also purchased Word Perfect at the same time and it still loads, no problem. I realize Microsoft cannot support it forever ever BUT I cannot even activate it!!!!!!!!!!!! (run it 50 times max). - Is Microsoft trying to drive the world away from their products, looking at the new Wordperfect X6 pro, it looks better for the money. 
Jul 29, 2003. Re: cynjar (11:29am est tue jul 29 2003) your system doesn't have to be hooked up to the internet to get authorized. If you have no 'net connection, ms provides a phone number for you to call in and get your authorization code. If you have no 'net connection and no phone, you're just sol. I have purchased a copy of Office 2003 from a seller on Ebay who states that they have uninstalled it on their PC, but when I try to activate I get a message that it.  
I hated Excel/Access. Outlook Express is not even in their products any more. Now Win 8 has NO DVD player, who watches DVD's on a computer beside Gamers/Geeks and my mother, my brothers and my sisters, friends etc. We wire our PC's to our huge flat screen TV's and don't need a separate DVD Player. MS should re-evaluate DVD sales numbers, there still there. 
I also have some business that wire to a wall mounted flat screen in their office's and meeting rooms and need DVD support for product reviews etc.(you don't post propriety or secure info on a web site or FTP, DVD's are one shot and you can carry them with you). - Linux is also looking good, the learning curve is much smaller than Window 8 and I find that Open Office and Thunderbird/Calendar while not perfect (what is) are a viable option. My suggestion to Business now is to get your IT guys to evaluate the option of Linux and other non MS products to see if they are viable for your situation. MGJ Smith just another unhappy MS Customer.. I, too, called the regular Tech Support person first, only to be told that I needed to have 'premium' technical support for $99. He would not give me the telephone number for activation (which, as you said, no longer appears on the activation screen). Much thanks to Palcouk for his solution, especially 'Do not respond to any prompts until you reach a live human.' The living person at the activation center helped me quite willingly. I've been working on this problem for several days. Glad it is finally resolved. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
March 2018
Categories |
 RSS Feed
RSS Feed
